Cobalt-catalyzed alkyne silylamidation unlocks Z-selective unnatural dehydropeptides synthesis
Jixin Wang1, Ting Zeng1, Kaixin Chen2, Zexu Chen1, Long Lin1, Wenhua Yu3, Jianlin Yao(姚建林)1*, Hong Yi(易红)2* , Baosheng Wei(魏保生)3* , Jie Li(李杰)1,4,5*
1State Key Laboratory of Bioinspired Interfacial Materials Science, College of Chemistry, Chemical Engineering and Materials Science, Soochow University, Suzhou 215123, China
2College of Chemistry and Molecular Sciences, The Institute for Advanced Studies (IAS), Wuhan
University, Wuhan 430072, China
3College of Chemistry and Chemical Engineering, Central South University, Changsha 410083,
China
4Suzhou Key Laboratory of Pathogen Bioscience and Anti-infective Medicine, Soochow University, Suzhou 215123, China
5MOE Key Laboratory of Geriatric Diseases and Immunology, Soochow University, Suzhou 215123, China
Natl. Sci. Rev.,2026,13, 5, nwag011
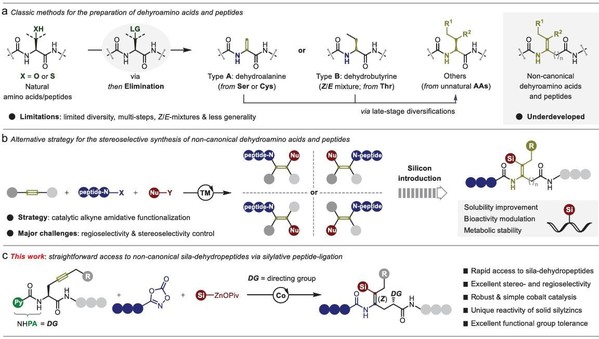
Abstract: Due to their unique conformational properties and activities, dehydropeptides play a pivotal role in the fields of biological and medicinal chemistry. Yet, the synthesis of unnatural dehydropeptides still suffers from cumbersome steps and less generality, in particular, with rather limited structural diversity. Herein, a modular cobalt-catalyzed 1,2-silylamidation of non-conjugated alkynes with dioxazolones and silylzinc pivalates is disclosed, thus affording structurally diverse non-canonical sila-dehydropeptides with complete control of regio- and stereoselectivity. Notably, the reaction enables efficient peptide ligation between peptide-containing dioxazolones and peptide-containing alkynes in a Z/E-stereoselective and diastereoretentive manner. Moreover, broad substrate scope, outstanding functional group compatibility, as well as facile late-stage diversifications of pharmaceutically active molecules substantiate the synthetic value of this method.
Article information: https://doi.org/10.1093/nsr/nwag011
